Gannex, a wholly-owned company of Ascletis,is fully dedicated to the R&D and commercialization of new drugs in the field of NASH. Gannex has two clinical stage drug candidates against two different targets – FASN, THR-beta .
ASC40, a Global First-in-class, Oral FASN Inhibitor for NASH
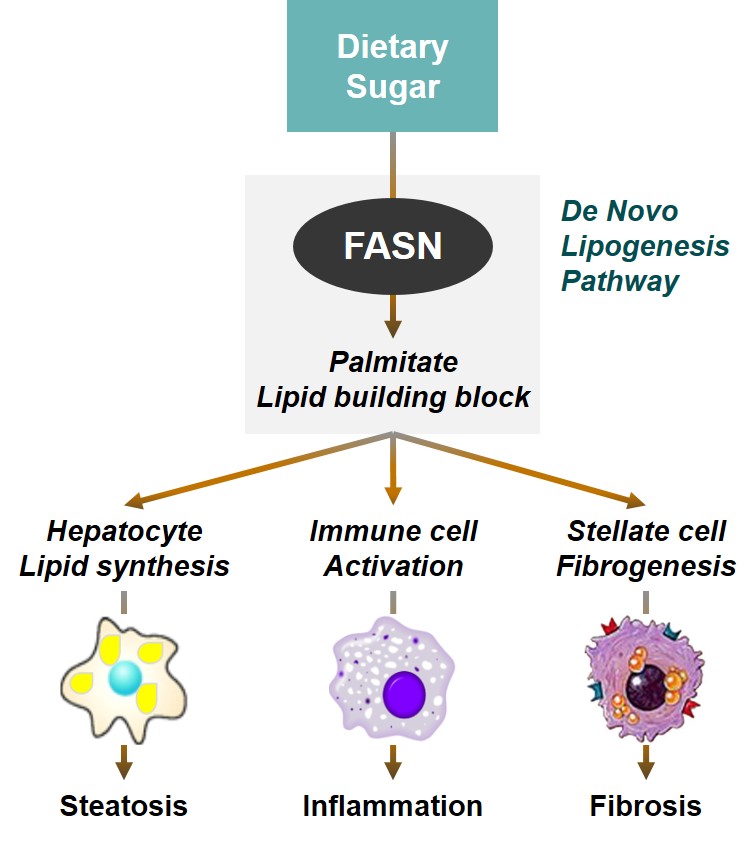
FASN: fatty acid synthase NASH: non-alcoholic
steatohepatitis NAFLD: non-alcoholic fatty liver disease
C. Estes et al., J HEP 2018 (69): 896–904
Efficacy and Safety of ASC40 Treatment in Patients with NASH
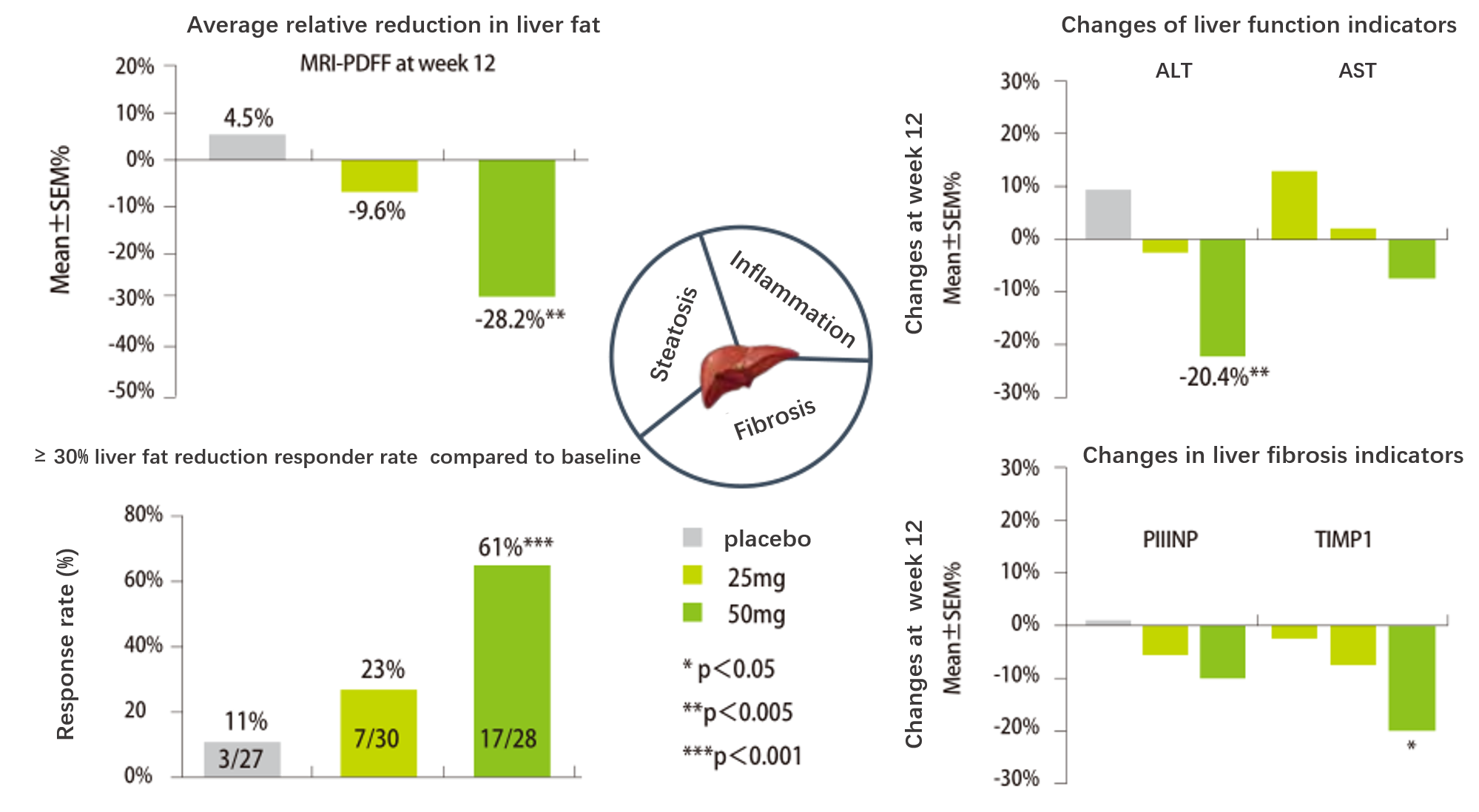
Rohit Loomba et al. 2020, Hepatology 72; 103.EASL 2020 Oral Presentation
ASC41, an Oral THR-β Agonist for NASH
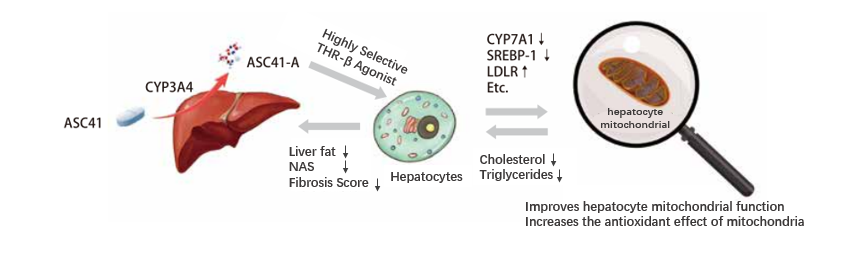
• Liver-targeted prodrug (ASC41) and active moiety (ASC41-A) is selective for THR-β
• In two NASH animal models, at 1/10 dose of MGL-3196, ASC41 demonstrated the same improvement in liver steatosis, inflammation and fibrosis
• A highly potent and selective THR-β agonist with anticipated human efficacious dose <10 mg QD
• Proprietary oral tablet formulation stable at room temperature and whose exposure is same as solution formulation in dogs
• Topline data of Phase I safety, PK and preliminary efficacy (LDL-C) in healthy volunteers with LDL-C > 110 mg/dL is expected to be available by the end of 2020
• Combination opportunities with ASC42 (FXR) and ASC40 (FASN)
Phase I Clinical Trial Results of ASC41
• 65 subjects with elevated low-density lipoprotein cholesterol (LDL-C) (> 110 mg/dL), a population characteristic of non-alcoholic fatty liver disease (NAFLD)
• In the single-ascending dose portion of the study, preliminary data suggest that ASC41 is safe and well tolerated up to a dose of 20 mg. Furthermore, ASC41 tablet formulation showed a dose-proportional pharmacokinetic profile from 1 mg to 20 mg
• In the multiple-ascending dose (MAD) portion of the study, preliminary data suggest that after 14 days of once daily oral dosing, subjects demonstrate clinically meaningful and statistically significant reduction in LDL-C and triglycerides compared to placebo, as shown in the table below:

• ASC41 had a benign adverse event profile at all doses following 14-day treatment, with no grade 3 or above adverse events, no serious adverse events or premature discontinuations
• ASC41 tablet formulation displayed a dose-proportional pharmacokinetic profile from 1 mg to 5 mg following once daily, 14-day dosing
Publications
Title | Venue | Type | Date |
| ASC41, a selective THRβ agonist significantly reduces liver fat and ALT in biopsy-confirmed MASH patients after 12-week treatment: an interim analysis of a 52-week serial liver biopsy study | The International Liver Congress™ 2024 of the European Association for the Study of the Liver (EASL) | Poster | 6/2024 |
ASC41, a thyroid hormone receptor β agonist, showed little drug interaction, significant lipid reduction and comparable pharmacokinetic profiles among Chinese and US healthy subjects and patients with non-alcoholic fatty liver disease (NAFLD): results from two phase 1 studies | The Liver Meeting® 2023 of the American Association for the Study of Liver Diseases (AASLD)
| Poster | 11/2023 |
| A Phase Ib Study to Evaluate the Safety, Tolerability and Pharmacokinetics of ASC 41 a THR-β Agonist, for 28-days in Overweight and Obese Subjects with Elevated LDL-C, a Population with Characteristic s Of NAFLD | The Liver Meeting® 2021 of the American Association for the Study of Liver Diseases (AASLD) | Poster | 11/2021 |
| Significant lipid lowering by ASC41, an oral tablet, liver-targeted THRβ agonist, in a phase I randomized, double-blind, placebo controlled single- and multiple-ascending dose study | The International Liver Congress™ 2021 of the European Association for the Study of the Liver (EASL) | Poster | 04/2021 |
| Significant Improvement of NAFLD Activity Scores and Liver Fibrosis by ASC41, a Selective THR-β Agonist, in High Fat Diet Induced NASH SD Rats | The International Liver Congress™ 2021 of the European Association for the Study of the Liver (EASL) | Poster | 04/2021 |
| Novel, first-in-class, fatty acid synthase inhibitor, TVB-2640 versus placebo demonstrates clinically significant reduction in liver fat by MRI-PDFF in NASH | The Liver Meeting® 2020 of the American Association for the Study of Liver Diseases (AASLD) | Oral | 11/2020 |
| Novel, first-in-class, fatty acid synthase inhibitor, TVB-2640 versus placebo demonstrates clinically significant reduction in liver fat by MRI-PDFF in NASH | The Liver Meeting® 2020 of the American Association for the Study of Liver Diseases (AASLD) | Oral | 08/2020 |
| The FASN inhibitor TVB-2640 is efficacious in a new 3D human liver microtissue model of NASH | The Liver Meeting® 2020 of the American Association for the Study of Liver Diseases (AASLD) | Poster | 08/2020 |
| Fatty Acid Synthase Inhibitor TVB-2640 Reduces Hepatic de Novo Lipogenesis in Males With Metabolic Abnormalities | Hepatology 2020 | Paper | 07/2020 |
| Progressive Reductions in Hepatic DNL with Increasing Doses of TVB-2640, a First-in-Class Pharmacologic Inhibitor of FASN | Keystone Symposium on Organ Crosstalk in Obesity and NAFLD | Poster | 01/2018 |
| Establishing the foundation for a novel, first-in-class, fatty acid synthase inhibitor, TVB-2640, for the treatment of NASH | The International Liver Congress™ 2017 of the European Association for the Study of the Liver (EASL) | Poster | 04/2017 |



