ASC06
2018年8月10日,歌礼合作伙伴、全球领先的核酸干扰技术(RNA interference,RNAi)疗法开发公司Alnylam Pharmaceuticals创新药物Onpattro (patisiran) 正式获美国FDA批准,这是全球第一个获得批准上市的利用RNAi技术开发的药物,代表着RNAi药物时代的到来。
歌礼旨在将ASC06开发成第一个通过使用RNA干扰技术系统性给药的肝癌治疗药物,其设计为抑制对癌细胞生长及发育至关重要的两个基因:VEGF及KSP。ASC06已完成I期及I期扩展临床试验,临床结果显示50%患者病情趋于稳定,1名患者获得完全应答。ASC06安全且患者耐受良好。歌礼预计于2020年在中国启动ASC06 II期临床试验。
当前疗法及局限
肝癌有多种治疗方案。当前肝癌的主要疗法包括系统化疗及靶向小分子药物。此类疗法的临床功效较低且副作用很大。因此,此类方案均未被视为有效治疗方案。尤其是针对晚期肝癌,目前尚无任何有效系统疗法。
ASC06 的优势根据已完成的临床试验及临床前研究,我们认为ASC06有潜力从以下方面解决当前肝癌疗法的局限性:
• 对适应症的疗效 ASC06的临床试验已表明其在肝脏的参与下对晚期实体瘤患者的初步活性。一名患有转移性肝癌的患者实现完全应答。总共24名患者中的12名(50%)接受≥0.7mg/kg剂量治疗,在两个月评估结束后患者病情趋于稳定,相较于≤0.4 mg/kg剂量治疗,12名患者只有1人病情趋于稳定。我们认为,ASC06具有成为肝癌首选疗法的巨大潜力。
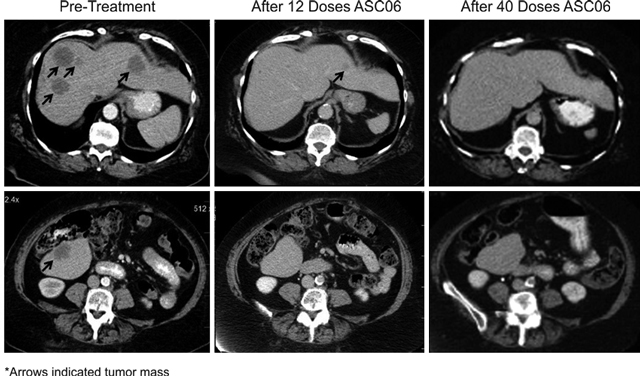
• 良好的安全性及耐受性 I期临床试验患者出现的大多数副作用的严重程度为1级或2级。此外,7名患者的平均治疗周期为11.5个月(包括I期),并无新毒性报告,这表明 ASC06具有良好的安全性并允许长期给药。我们认为,良好的安全性及耐受性特征将允许更多的患者接受长期治疗。